|
What an amazing experience! I feel so grateful for being given the opportunity to take the C-MORE summer microbial oceanography course. When I reflect on the past 5 weeks it is incredible how much was covered in the lectures, research cruise and lab work. But perhaps the most valuable aspect of the course was the connections made working with the other C-MORE students and the faculty of the course. We came from from very diverse backgrounds but we all share a passion for better understanding the intriguing world of the very small in the ocean. I feel truly inspired to apply what I learned into my own research and in the future I hope to form collaborative projects with these bright and amazing scientists. Mahalo!
2 Comments
 Measuring low level nutrients on a spectrophotometer Measuring low level nutrients on a spectrophotometer After ten days of research at sea on the cruise we brought back a ton of samples to analyze in the C-MORE labs. We measured nutrient concentrations, primary and bacterial production, chlorophyll a concentrations, ATP and particulate carbon, nitrogen and phosphorus. We spent the last week of the course running these samples in the lab and analyzing the data. We used these data along with data collected from the CTD, optical sensors and the genomic samples to make a 2 hour presentation on our findings. So much data! 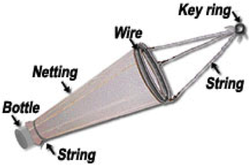 http://www.bigelow.org/foodweb/satellite2.html http://www.bigelow.org/foodweb/satellite2.html The most abundant forms of life at Station Aloha are those that cannot be seen by the naked eye. Bacteria, viruses, and most phytoplankton and zooplankton can only be seen using a microscope. In order to view the plankton in the water you can use a funnel shaped net called a plankton net which is dragged or "towed" through the water. The net allows water and very small particles flow through but collects plankton that are bigger than the mesh size. The plankton are concentrated in a bottle attached to the bottom of the net. The contents of the bottle (lots of phyto and zoo plankton!) can then be viewed under a microscope. We saw pennate and centric diatoms, radiolaria, copepods and much more. 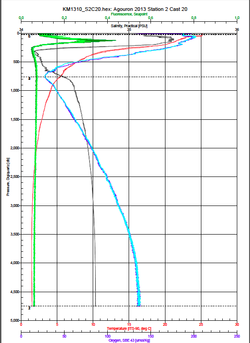 Deep CTD cast on 6.19.2013. Deep CTD cast on 6.19.2013. Station Aloha is approximately 4800 meters deep. During the research cruise we took 2 CTD casts from the surface waters down to the very bottom. The deep ocean water is cold and salty making it very dense. At the surface the water is a balmy 25 degrees Celsius (77 degrees Fahrenheit) while at depth it is near freezing temperatures! The deep waters also have high oxygen because the deep ocean water comes from the cold, oxygen-rich deep waters from the poles. The water is transported by large ocean currents that connect all oceans. This is called thermohaline circulation. The movement of water is driven by density differences and is sometimes described as the "great ocean conveyer belt". 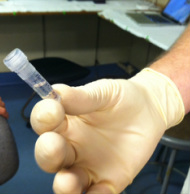 Gold! Gold! Hard to believe but this tiny bottle contains genetic information that will allow us to characterize the diversity of the microbial community at Station ALOHA. This was the final product from 5 days of sampling and many hours filtering, extracting, priming, amplifying and pipetting! We took water samples from the surface of the water all the way through the water column. We are interested in determining the diversity of microbial organisms at Station Aloha. We used PCR (polymerase chain reaction) to amplify pieces of DNA across several orders of magnitude. Each sample received a specific primer which serves as "tag" so we can link the microbial community to a depth and time of day they were collected. Primers are short DNA fragments that contain sequences complementary to the target region along the DNA polymerase. We checked to see whether the PCR generated the anticipated DNA fragments (sometimes called amplicons) using a process called agarose gel electrophoresis, which separates the PCR products based on size. A DNA ladder containing DNA fragments of known sizes is run alongside the PCR products. Once we determined that the PCR worked for each sample we then conducted a procedure that pooled all of the samples together and we ended up with one small bottle. It is truly mind blowing to consider how much data and information is contained in such a small volume! 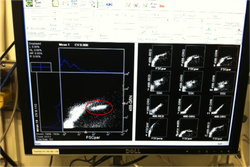 Prochlorococcus community circled in red. Prochlorococcus community circled in red. Flow cytometry is a laser based biophysical technology used for cell counting and sorting. It is used widely in the medical field to analyze blood samples but over the past few decades it has become an important instrument used in marine biology to determine microbial community structure and abundances. Flow cytometry suspends cells in a stream of fluid that then passes by an electronic detector. The analysis can characterize the physical and chemical attributes of up to thousands of particles per second. At Station Aloha and other low nutrient regions of the ocean, a genus of photosynthetic microorganisms called prochlorococcus are abundant and can be identified and quantified using flow cytometry (see picture). Prochlorococcus are incredibly small (0.6um) cyanobacteria that are the most abundant photosynthetic organisms on earth. One milliliter of seawater may contain up to 100,000 cells of these organisms. Due to their small size Prochlorococcus have a high surface to volume ratio which gives them a competitive advantage in low nutrient waters. They can also grow at very low light conditions. Despite their ecological importance they were not discovered until 1986 by Sallie W. (Penny) Chisholm (Massachusetts Institute of Technology) and other collaborators. The discovery was made using flow cytometry methods in the Sargasso Sea.  SQF getting ready to recover the array. SQF getting ready to recover the array. Thursday was an awesome day! I love working on the deck. For the whole day our group was responsible for the deployment and recovery of everything put into the water. This included all of the CTD/niskin rosette casts, the productivity array, and optical instrument casts. The day started at 11:30pm (the night before)! Our first cast was at midnight and it included 3 different optical instruments plus a small CTD that were attached to the same metal cage. The optical instruments included a LISST (Laser In Situ Scattering Transmissometry), AC9, and ACplus. These are active optical instruments which means that they analyze the optical properties of the water using laser and light rays opposed to using light from the sun. This is why we can do the cast in the middle of the night. We also deployed a productivity array which contained bottles of sea water spiked with the radio isotope 14C and other bottles spiked with tritiated leucine (an amino acid with the radio isotope 3H). These bottles were placed on a line at different depths and then put into the water at sunrise and then floated around attached to buoys and an Argos transmitter. At sunset we tracked down the array and hauled it back on deck. The samples were then analyzed to determine the rate of CO2 uptake (primary productivity) using the 14C label and also the rate of protein synthesis /bacterial productivity using the tritiated leucine label.  CTD and niskin rosette. CTD and niskin rosette. Aloha! Today is Saturday 6.15.2013, day 5 of our research cruise. It has been an exciting week of science! Due to slow/inconsistent internet it has been difficult to update this blog and we have done a lot so I have tons to write about! On Wednesday 6.12.2013 my student group ("The Kraken", self-named:) worked on analyzing water samples collected at multiple depths between the surface of the water down to 1000 meters using the niskin sampling tubes on the CTD rosette (see picture). We took samples to measure important basic water quality parameters such as dissolved inorganic nutrients, dissolved oxygen, particulate carbon, nitrogen and phosphorous, and ATP. The ATP analysis was particularly interesting. ATP (Adenosine-5' triphosphate), is a coenzyme that transports energy within cells for metabolism. It can be thought of as the "molecular unit of currency". Since ATP is found in and around all living organisms, measuring the total concentration (amount per volume) of ATP in our water samples provides a measure of total microbial biomass. The analysis for quantifying ATP is super cool...After filtering our water sample onto a small filter, we place it into a tube filled with a boiling base solution. This step breaks up the cells and extracts the ATP. After the extraction we add "firefly dust" which contains an enzyme (luciferase) and subtrate (luciferin) extracted from fireflies! The reaction produces light and the amount of light produced is directly proportional to the amount of ATP and therefore the amount of living organisms in the sample! The reaction is catalyzed by the enzyme (luciferase) simplified to this: luciferin + ATP --> oxyluciferin + AMP + light |
Sarah Q. FosterSarah is a 2nd year Ph.D. student in the Fulweiler Lab. This blog documents her experience taking a summer course "Microbial Oceanography: From genome to biome" at C-MORE at the University of Hawai'i at Manoa. ArchivesCategories |
















 RSS Feed
RSS Feed